Press release2022.01.25
AI-based software supports efficient healthcare
New technology is paving the way for major improvements in the healthcare sector. One example is the AI-based operation system from medtech company Ortoma that enhances quality and streamlines the process for hip joint, knee joint and back surgery. In a new collaboration, Semcon is supporting Ortoma with specialist skills in software development and technical information.
This collaboration is a great example of how digital solutions really help people and it clearly shows how we can contribute to innovative solutions through our assignments at Semcon.
– Oskar Åkerlund, Team Manager, Software & Emerging Tech at Semcon
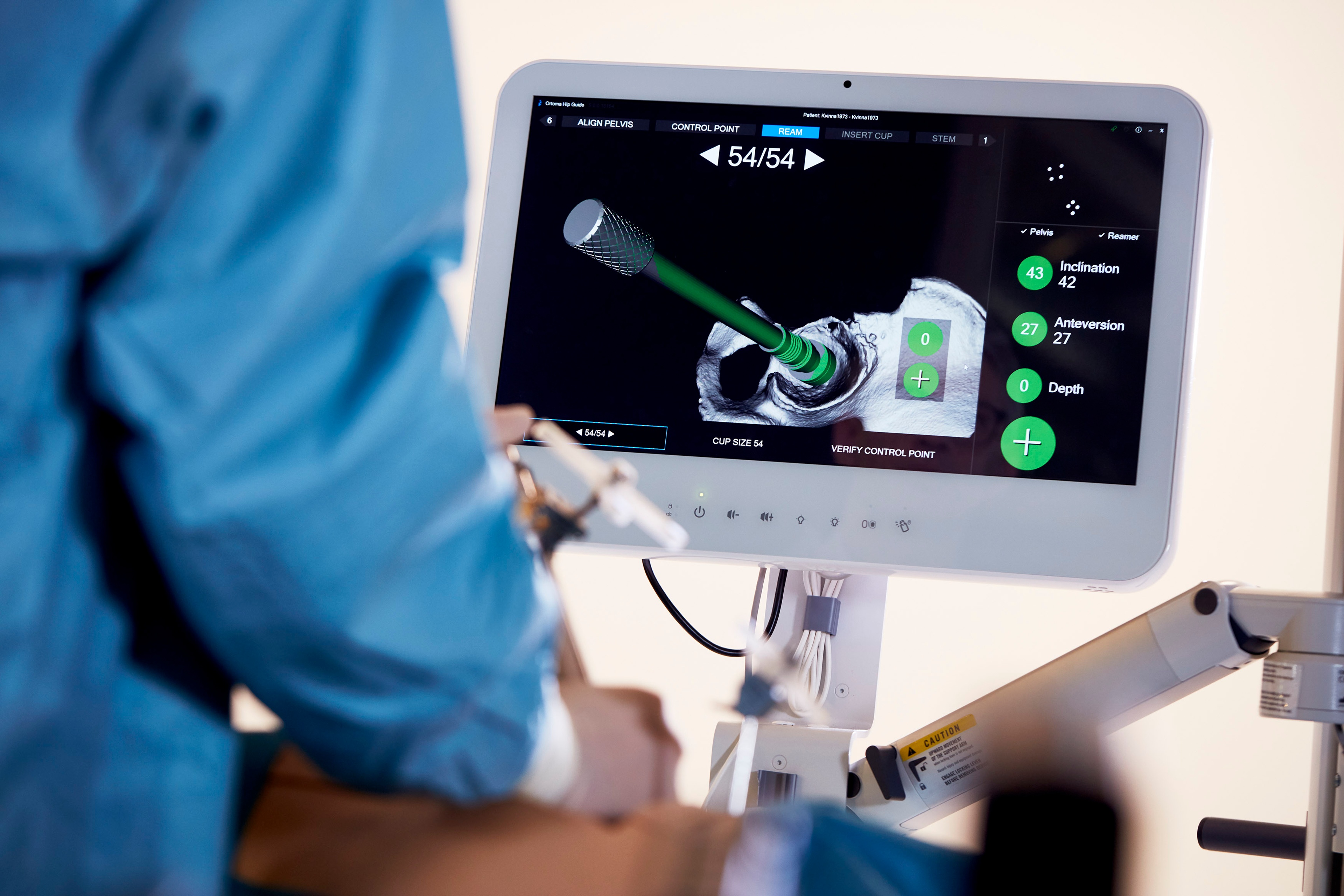
Using the Ortoma Treatment Solution (OTSTM) system allows surgeons to plan their operations in 3D, including the ability to identify optimal positioning for an implant in the patient. OTS can also be used for navigation during the operation, as well as support in the post-operative medical follow-up of a patient.
“Ortoma is an exciting new customer for us at Semcon and we look forward to supporting the company in the development of systems with artificial intelligence in order to support enhanced and streamlined surgical procedures”, says Oskar Åkerlund.
Digital solutions that deliver benefits
Semcon and Ortoma launched their collaboration towards the end of 2021 and have also signed a framework agreement that runs until further notice and includes specialist skills in software development and technical product information.
"By working with Semcon we are increasing our access to specialist skills and we look forward to continuing the development of our AI-based software. OTS is a system that automates many work steps that would otherwise be performed manually by surgeons, and does so in a fraction of the time it takes for conventional systems." says Linus Byström, CEO of Ortoma
About Ortoma
Ortoma was established in 2002 with the objective of developing products to improve people’s lives. The company focuses on the development of solutions for orthopaedic operations and has regulatory approval for products in Europe, the USA and Japan. The company has been listed on the Spotlight Stock Market since 2014.

